Contents1. Why cold runners drive waste in medical molding2. Eliminating Cold Runner Waste with hot ...
Eliminating Cold Runner Waste: The Benefits of Hot Runner Injection Molding
Contents
1. Why cold runners drive waste in medical molding
2. Eliminating Cold Runner Waste with hot runners: mechanisms and material notes
3. 12‑month ROI and TCO: method and worked example
4. Retrofit checklist: 10 practical steps
5. Validation for cleanrooms: what quality will ask for
6. Process control best practices (with a neutral micro‑example
7. Risks and practical mitigations

Medical consumables molders working with PP and PC fight a familiar battle: every cycle produces parts—and an equally predictable mass of cold runner. When regrind is restricted or banned (as it often is in cleanrooms), that runner is pure waste. In multi-cavity tools, runners can account for a large share of the shot, so switching to a hot runner can conservatively remove 20–40% of material consumption tied to the runner alone. Think of it this way: if the runner weighs almost as much as the parts, melting it every shot is like buying material to make nothing. For teams focused on Eliminating Cold Runner Waste without compromising validation rigor, hot runners are often the most direct lever.
This best-practice guide walks through how hot runners eliminate that waste, what changes in cycle time and quality you can reasonably expect, and how to plan a compliant retrofit for medical PP/PC production. Our running example is a small medical connector molded in PP or PC inside an ISO-classified room. Along the way, you’ll find a reproducible 12‑month ROI method, a retrofit checklist, and the validation steps your quality team will expect.
Why cold runners drive waste in medical molding
Cold runners deliver the melt, then solidify and exit with the parts. In consumer applications, some percentage may be reground and blended back. In medical manufacturing, the default is different: many customers specify virgin-only resin or tightly limited regrind to avoid contamination, property drift, and traceability issues. That policy makes the runner mass per shot a direct operating cost and underscores why Eliminating Cold Runner Waste is financially material in cleanrooms.
Industry references illustrate how runner mass can significantly impact shot size in multi-cavity molds—reinforcing why eliminating it can make a substantial difference. For background on shot sizing and the impact of runner geometry on total shot mass, see the discussion in Plastics Technology’s overview of calculating shot size versus barrel capacity, which shows how runner and sprue volume can be a significant portion of each shot in practice (2017) in the article on a simpler way to calculate shot size vs. barrel capacity: Plastics Technology’s shot-size method.
Eliminating Cold Runner Waste with hot runners: mechanisms and material notes
Hot runners keep the polymer molten inside an insulated manifold, heating the drops, so the runner no longer solidifies and ejects with the parts. Mechanistically, that removes the runner from each cycle and reduces the cooling time associated with thick runner sections. It also improves gate consistency and reduces shear history versus long, cooling runners—often leading to better dimensional stability and fewer cosmetic defects.
Gate strategy matters:
Open (thermal) gates are simpler and work well for many PP medical parts.
Valve gates add positive shutoff, which can help with PC’s sensitivity to stringing and gate vestige on cosmetic or functional faces.
Material notes for PP and PC:
PP typically runs with melt temperatures around 200–250°C and moderate mold temperatures; it’s tolerant but still benefits from uniform, low-shear gating.
PC generally wants 280–320°C melt, a warmer mold, strict drying, and tight residence-time control. Stable hot runner temperatures (±1–2°C at the drops) help avoid splay and yellowing while Eliminating Cold Runner Waste tied to solidified runners.
For system features and mechanisms, vendor handbooks outline how manifold design, drop selection, and valve pin actuation influence gate quality and cycle. A concise background is available in the 2021 Husky hot runner product handbook: Husky’s Hot Runner Product Handbook v18.1.
12‑month ROI and TCO: method and worked example
Here’s the deal: without audited public medical case studies listing exact percentages, the safest way to plan is to run your own arithmetic with conservative assumptions. Use these inputs:
Material cost ($/kg)
Runner-to-part mass ratio (baseline, cold runner)
Annual shots
Cycle-time change (seconds/cycle)
Machine rate ($/hr) and labor delta (if manual degating/handling goes away)
Energy delta (optional; usually smaller than material/time effects)
CAPEX (hot runner + controller + installation + downtime + validation)
Worked example (medical PP connector, 16 cavities):
Baseline shot: parts 48 g total; runner 16 g (runner = 25% of shot)
Annual shots: 4,000,000
PP price: $2.20/kg
Cycle-time reduction: 4 s per 20 s cycle (→ 20% faster)
Machine rate: $65/hr
CAPEX + validation: $210,000
Regrind allowed: 0% (cleanroom virgin-only policy)
Calculations in plain language:
Annual material saved = 4,000,000 shots × 0.016 kg/shot = 64,000 kg → $140,800/year
Time saved = 4 s × 4,000,000 / 3600 = 4,444 hr → $288,860/year at $65/hr
Total annual benefit (before minor adds/deducts) ≈ $429,660
Payback ≈ $210,000 / $429,660 ≈ 0.49 years (~6 months)
Sensitivity bands: If runner share is only 15% and cycle delta is 3 s, payback extends but typically remains within 12 months for high-volume tools. If your runner is 35% and cycle delta 6–8 s, payback can shorten considerably. A practical explanation of why cycles drop (no runner cooling/handling) is covered in MoldMaking Technology’s transitional guidance (2023): How to transition from a cold to hot runner mold.
ROI calculator snapshot (example numbers):
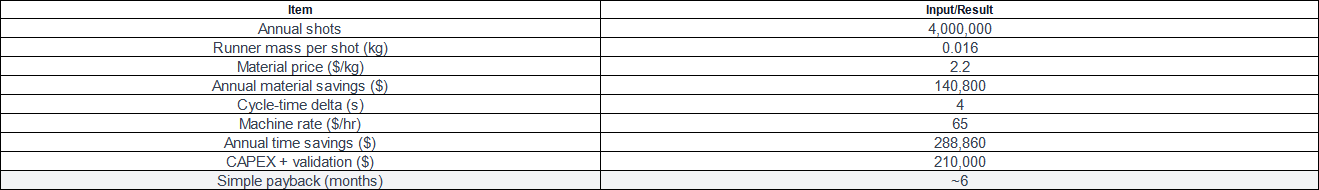
Note: Treat these as placeholders for your own values; the method, not the exact numbers, drives decisions. If you run PC at higher melt/mold temperatures, confirm energy impacts in your plant’s metering—material and time remain the primary levers. For SEO clarity and reader search intent, remember the objective here is Eliminating Cold Runner Waste while maintaining medical compliance.
Retrofit checklist: 10 practical steps
Before any hardware order, baseline your current state and plan change control. A cross‑functional team (molding, tooling, maintenance, quality, supplier) should own the following:
Mold survey and measurement: Weigh runner vs. parts per shot; document cavitation, gate types, and runner layout. Capture part CTQs and current scrap modes.
Manifold and gate selection: Choose open vs. valve gate based on resin (PP/PC), gate vestige needs, and cleanroom handling.
Controller requirements: Define zones, amperage, accuracy targets (aim for ±1–2°C at drops for PC), data logging/export needs, alarms, and communications to your MES/SCADA.
Utilities and safety: Plan heater/TC wiring, grounding, insulation, water layout, and press interfaces. Verify electrical loading and fusing.
Bench test and zone mapping: Continuity checks, TC polarity, soft‑start, and initial PID tuning. Record steady‑state stability at each setpoint.
Molding trials and DOE: Viscosity curve, gate freeze, fill/pack/cushion studies, and trim alarm thresholds from observed capability.
IQ documentation: Installation checks, calibration certs, wiring maps, interlock and alarm tests.
OQ protocols: Establish process windows with worst‑case runs; define acceptance criteria for dimensions and visual requirements.
PQ capability: Run at nominal settings; target Cpk ≥ 1.33 on CTQs, verify defect and particulate rates meet the validated limits.
Training and SOPs: Purging steps, color‑change protocol, valve‑pin service in cleanrooms, spare parts kit, and periodic TC/heater checks.
Validation for cleanrooms: what quality will ask for
A hot‑runner conversion is a controlled process change. Under ISO 13485, expect formal change control, risk assessment, and documented validation in your QMS. The FDA’s process validation lifecycle defines IQ, OQ, PQ and then continued process verification (CPV). If the press cell sits in an ISO 14644‑classified room, confirm that modifications do not degrade air cleanliness.
Two canonical references frame these expectations: FDA’s Process Validation Guidance (2011) and ISO 13485’s quality management requirements. For room classification context, see ISO 14644‑1’s particle‑based classes.
Validation essentials (build these into your plan):
IQ: Verify installation (heater/TC mapping, electrical loads, interlocks), controller calibration, alarm tests, and documentation (as‑built manifold layouts, zone maps).
OQ: Define process windows via DOE, including gate freeze and worst‑case extremes; log zone temperatures and alarms; set control limits.
PQ: Run capability lots at nominal; demonstrate stability (e.g., Cpk ≥ 1.33 for CTQs), confirm cosmetic and particulate limits; finalize inspection and CPV plans. As you execute these steps, keep the objective visible: Eliminating Cold Runner Waste while preserving validated quality.
Process control best practices (with a neutral micro‑example)
Stable thermal control turns a good manifold into a consistent process. Focus on:
Tight zone control and mapping: Confirm each thermocouple reads the intended drop and polarity is correct.
PID tuning: Autotune is a start; manual refinement often trims overshoot and hunting, especially for PC.
Data logging and alarms: Capture setpoint, actual, power %, and alarm events at 1–5 s intervals during trials; export to your QMS/MES for CPV.
Micro‑example (medical PC connector, 16 drops): A plant team installs a hot runner and maps 16 nozzle zones plus two manifold zones. During IQ, they discovered two thermocouples swapped—caught by a zone “bump” test and corrected before OQ. OQ begins with a 300°C PC setpoint at the drops and 100°C mold temperature. Autotune yields a 2.5°C overshoot and slow settling on four nozzles feeding thinner walls. The team narrows integral time and increases derivative slightly on those zones, reducing steady‑state variance from ±2.4°C to ±1.1°C at the drops. They also set alarms: over‑temp at +8°C, under‑temp at −10°C for startup, and load fault detection. This attention to thermal stability is central to Eliminating Cold Runner Waste without introducing new variability.
To support this workflow, a temperature controller with clear zone mapping, PID adjustment, and data export simplifies both OQ and CPV. As one neutral example, Topower supplies hot runner temperature controllers used in multi‑zone medical tools; engineers typically configure zones to match each drop and record trend data during validation. You can review the company’s general offering here: Topower Electronic Technology Co., Ltd. The point isn’t the brand—it’s ensuring your chosen controller provides accurate, stable control, audit‑friendly logging, and alarm traceability.
Risks and practical mitigations
Dead spots and degradation: Avoid stagnant volumes; work with the supplier to optimize manifold balance and purging procedures.
Color changeovers: Define a purge compound and temperature ramp; document steps to minimize hang‑up and cross‑contamination.
Gate wear and stringing: For PC, consider valve gates and verify shutoff timing; inspect pins and bushings on a preventive schedule.
What to do next
If you’re evaluating a cold‑to‑hot conversion for a PP or PC medical consumable, run your numbers with the ROI method above and walk the retrofit and validation checklists with your cross‑functional team. When you’re ready, request our neutral retrofit selection checklist and a sample ROI spreadsheet so you can plug in your own runner mass, cycle deltas, and costs. It’s the fastest path to Eliminating Cold Runner Waste with numbers your finance team can trust.
A final note on expectations: external references consistently explain why eliminating runners cuts waste and can shorten cycles, but every mold is unique. Use conservative inputs, cite your assumptions, and validate performance under your QMS.